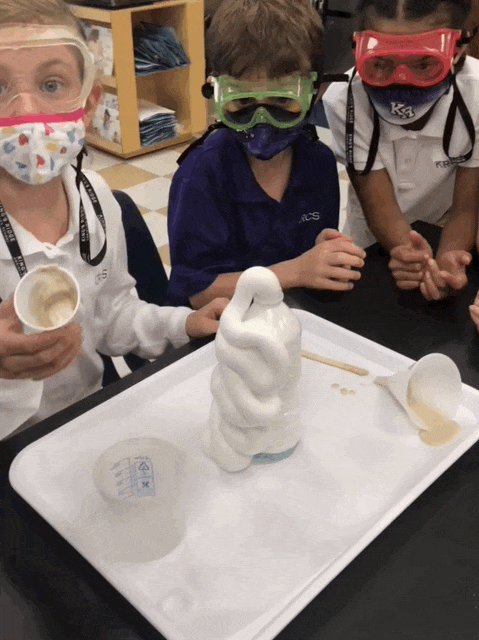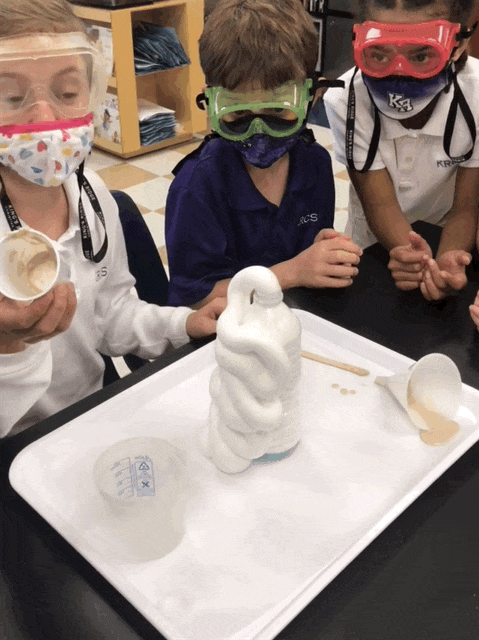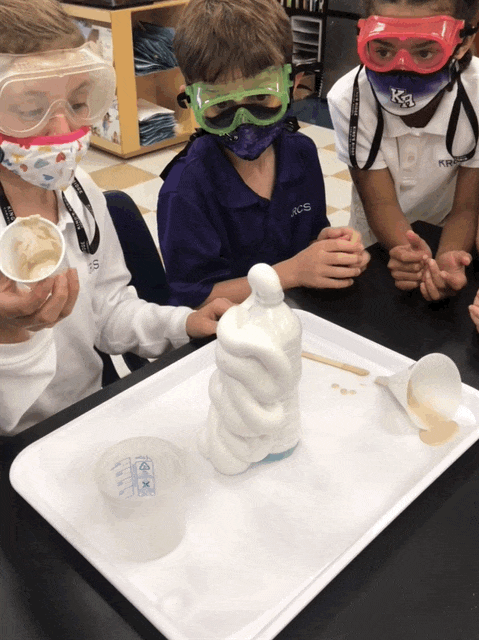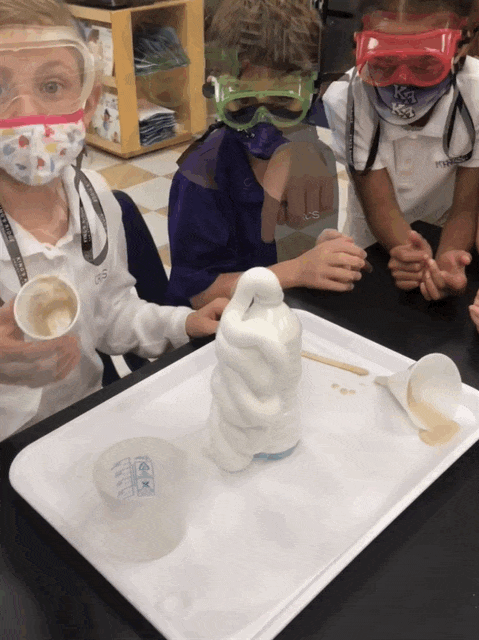
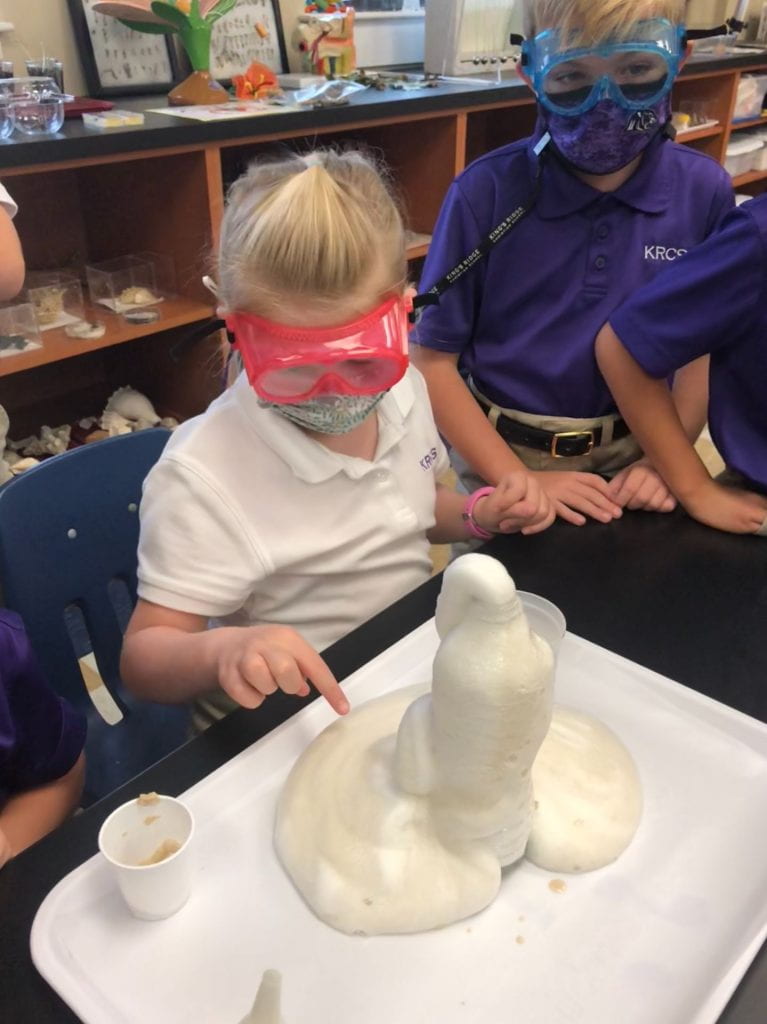
Elephant Toothpaste/Monster Foam
Second grade chemists created an exothermic (heat producing) chemical reaction when they mixed 1/2 cup of 20 volume hydrogen peroxide (purchased at Sally’s Beauty Store), a few squirts of Dawn dishwashing soap, and 1 T yeast dissolved in 50 mL of warm water. Yeast was our catalyst. This lab provided an excellent opportunity to review lab safety. I also introduced chemical formulas.
Before the experiment, we learned that yeast is a one-celled living organism that reproduces by budding. It is in a dormant state in the package, but we can “wake” it up with warm water. Yeast is used to make bread dough rise as carbon dioxide is released. Click here to watch an animated video of yeast budding.
In some of the bottles, we changed variables. We didn’t place soap in one and and in another, we used cold water, rather than hot to dissolve the yeast. We added food coloring in one bottle, and it really looked like toothpaste!
Click here or here for experiment directions.
Click here to watch a larger eruption!
We learned that unleavened bread does not have yeast in it and therefore does not rise. The Bible mentions yeast 31 times. That would be an interesting study!
Deuteronomy 16:3
“You shall not eat leavened bread with it; seven days you shall eat with it unleavened bread, the bread of affliction (for you came out of the land of Egypt in haste), so that you may remember all the days of your life the day when you came out of the land of Egypt.