Frost
What did I observe when I walked into school one cold morning?
What is the difference between frost and snow? How does frost form? Click here to watch this breathtaking beauty and click here to learn more about how and when frost forms.
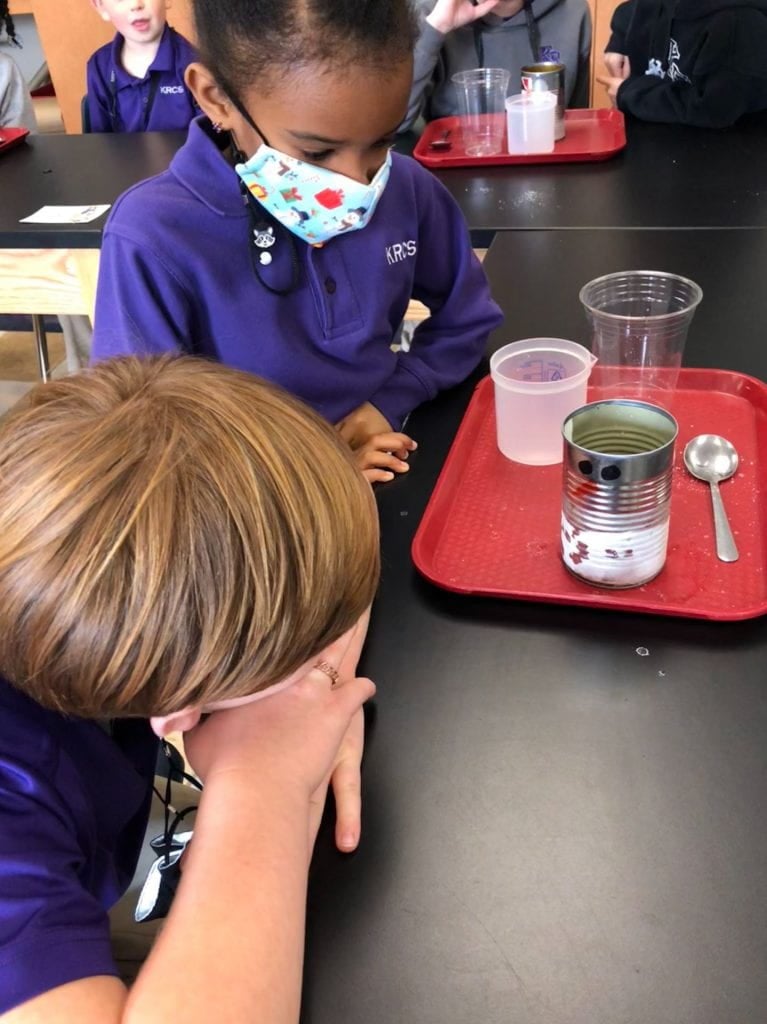
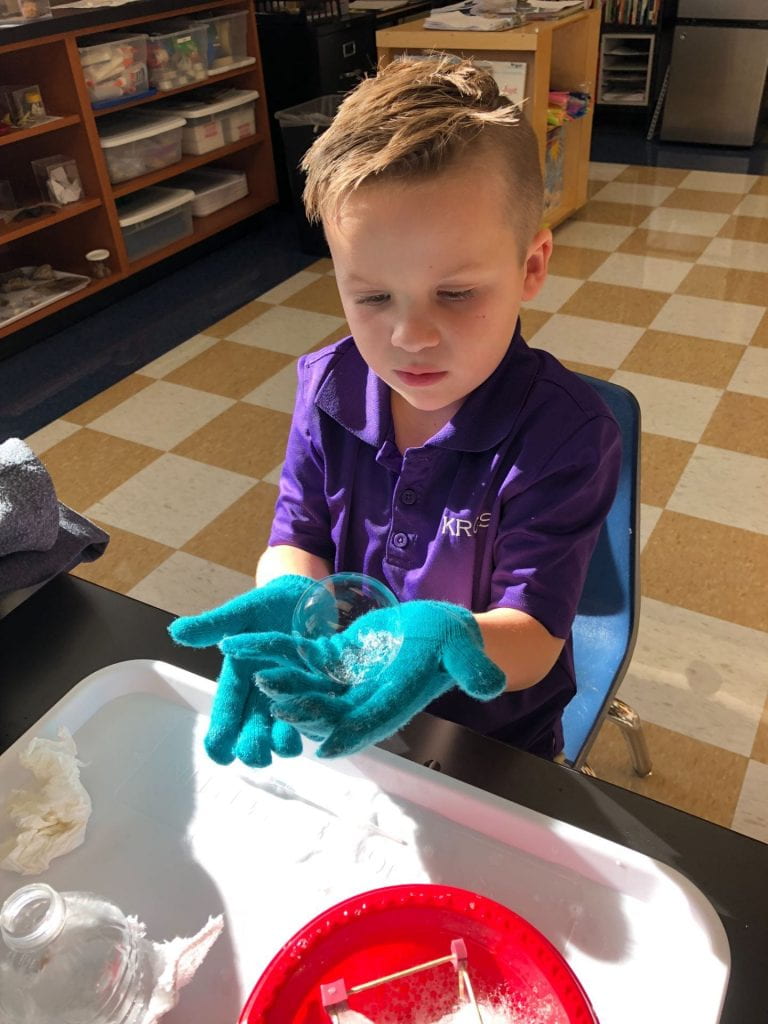
Can we make frost? Of course we can in the science lab! We poured 4 T of rock salt and 75 mL of water (from a beaker) into a recycled can that was filled approximately 2/3 of the way with ice. Then we chomped the ice about 20 seconds with a spoon. We were cautious not to touch the sides of the can because our hands are almost 99 degrees! We also did not place our hands inside the can because the edges of cans can be sharp. Wow, the spoon felt cold in the can! Why? Almost immediately, frost began to form on the outside of the can! But wait, we didn’t place anything on the outside of the can. Why did the ice crystals form there?
Frosty the Snowman
We briefly discussed different types of salt and where salt is found. During biblical times, salt was a valuable commodity, and it is mentioned over 40 times in the Bible. Investigating how it is used would be a great Bible study. We are called to be the salt of the earth. Matthew 5:13 What does that mean? I recommend this children’s book, if you want to learn more.
In this lab, there was a strong connection between math and science. We looked at a few types of thermometers and we learned freezing and boiling points.
On cold mornings, look for frost and observe the patterns. Try this experiment at home with different types of salt. How did your results differ?
While our frost was forming, we made crystals another way. We mixed 1 cup of Epsom salt (which actually isn’t salt, but magnesium sulfate) with 2 cups of boiling water. We poured the solution into Petri dishes. After the water evaporates, the crystals will be left.
He sends the snow like white wool; he scatters frost upon the ground like ashes. Psalm 147:16